 It turned out that the craftsman had been defrauding the King! Legend has it that Archimedes was so excited about his discovery that he ran naked through the streets of Sicily shouting Eureka! Eureka! (Which is Greek for “I have found it!”). He found that more water spilled over the sides of the tub when the craftsman’s crown was submerged. It differs by about 9, and therefore ice floats on liquid. It has a maximum density of 3.98 ☌ (1000 kg/m 3 ), whereas the density of ice is 917 kg/m 3. Water differs from most liquids in that it becomes less dense as it freezes. Then he placed the crown in a full container of water and the pure gold in a container of water. Pure water has its highest density of 1000 kg/m3 at a temperature of 3.98oC (39.2oF). Archimedes first weighed the crown and weighed out an equal mass of pure gold. 
He realized that a given mass of silver would occupy more space than an equivalent mass of gold. One day while entering a bath, he noticed that water spilled over the sides of the pool, and realized that the amount of water that spilled out was equal in volume to the space that his body occupied. Archimedes had no immediate answer and pondered this question for sometime. While you can round the density to 1 gram per milliliter, there are more precise values for you to use. The usual value used in calculations is 1 gram per milliliter (1 g/ml) or 1 gram per cubic centimeter (1 g/cm 3 ). The king challenged Archimedes to determine if the crown was pure gold. The density of water is the weight of the water per its unit volume, which depends on the temperature of the water. 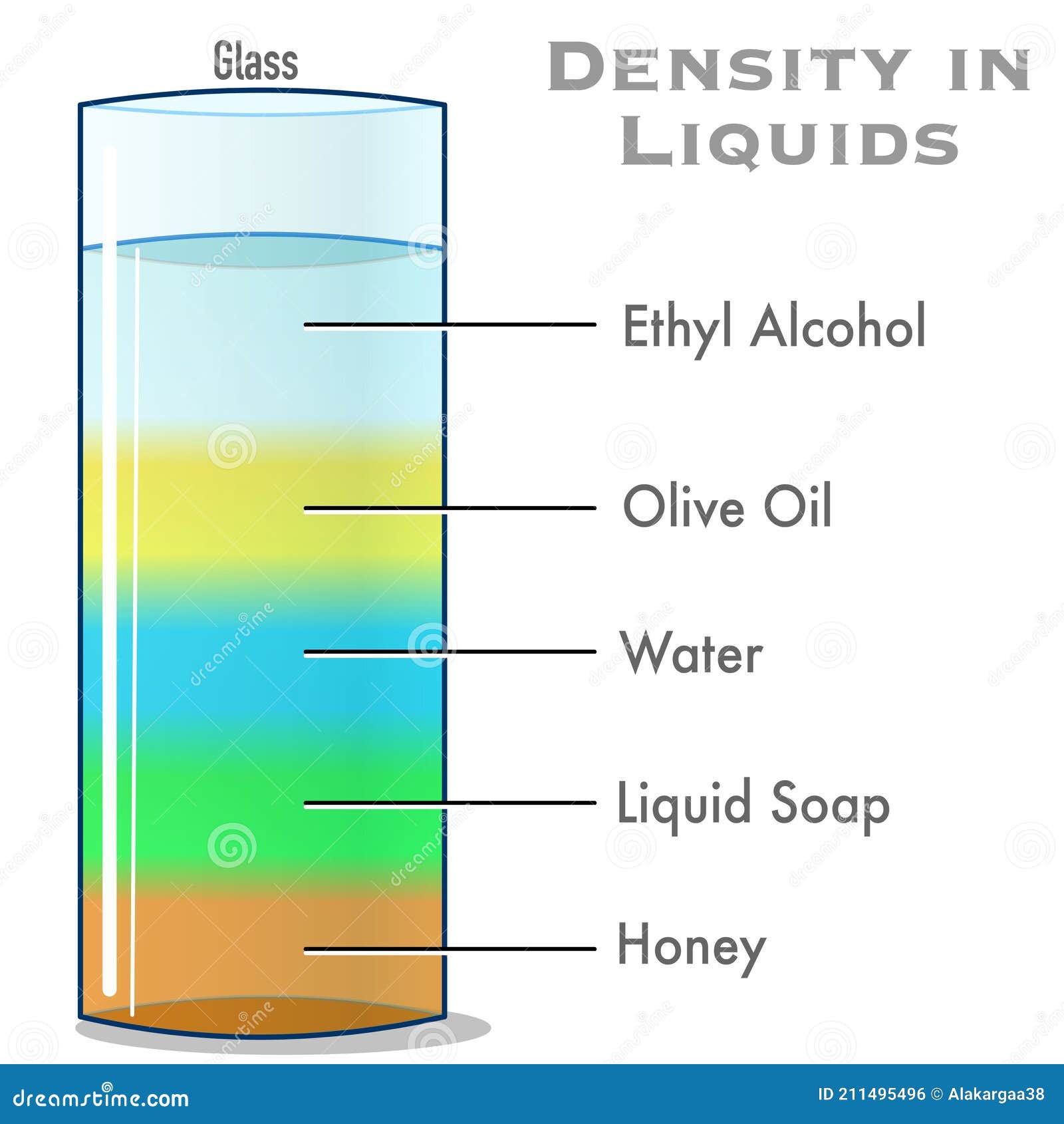
The crown weighed the same as other crowns but due to its intricate designs it was impossible to measure the exact volume of the crown so its density could be determined. The Specific Gravity of a substance is the ratio of its mass to that of an equal volume of water at the same temperature and pressure. The king suspected that the metal smith had added less valuable silver to crown and kept some of the gold for himself. The specific volume is the reciprocal of the density of the material, which is the mass per unit volume: r (1/v) (m/V). The king had provided a metal smith with gold to make a crown. Sometime around 250 B.C., the Greek mathematician Archimedes was given the task of determining whether a craftsman had defrauded King Heiro II of Syracuse. The discovery of specific gravity makes for an interesting story. Note that since water has a density of 1 g/cm3, the specific gravity is the same as the density of the material measured in g/cm3. Specific gravity values for a few common substances are: Au, 19.3 mercury, 13.6 alcohol, 0.7893 benzene, 0.8786. Similarly, an object with specific gravity less than 1 will float and those with a specific gravity greater than one will sink. An object will float in water if its density is less than the density of water and sink if its density is greater that that of water. Since specific gravity is a ratio, so it has no units. At this temperature the density of water is at its greatest value and equal 1 g/mL. Specific gravity is the ratio of density of a substance compared to the density of fresh water at 4☌ (39° F). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
